Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Sasaki, Yuji; Suzuki, Hideya*; Sugo, Yumi; Kimura, Takaumi; Choppin, G. R.*
Chemistry Letters, 35(3), p.256 - 257, 2006/03
Times Cited Count:36 Percentile:68.07(Chemistry, Multidisciplinary)Novel water-soluble organic ligands, N,N,N',N'-tetramethyl-diglycolamide (TMDGA), N,N,N',N'-tetraethyl-diglycolamide (TEDGA), N,N,N',N'-tetrapropyl-diglycolamide (TPDGA) and N,N-dipropyl-diglycolamic acid (DPDGAc), were synthesized and examined for complexation with An(III) and An(IV). These reagents are readily soluble in water and form more stable complexes with Pu(IV) and Am(III) than does EDTA of hexadentate ligand. TEDGA and TPDGA can back-extract effectively An into the aqueous phase. In addition, the An-complex in the aqueous phase can be dissociated and the TEDGA can be separated from the An and recovered by extraction using a polar organic solvent.
 -tetraoctyl-3-oxapentanediamide (TODGA) from nitric acid into
-tetraoctyl-3-oxapentanediamide (TODGA) from nitric acid into  -dodecane
-dodecaneZhu, Z.-X.; Sasaki, Yuji; Suzuki, Hideya*; Suzuki, Shinichi; Kimura, Takaumi
Analytica Chimica Acta, 527(2), p.163 - 168, 2004/12
Times Cited Count:263 Percentile:99.08(Chemistry, Analytical)The extraction of 75 elements by  -tetraoctyl-3-oxapentanediamide (TODGA) from nitric acid to
-tetraoctyl-3-oxapentanediamide (TODGA) from nitric acid to  -dodecane was investigated and discussed concerning distribution ratios (D) of metal ions and their ionic radii. The extractability for elements depends strongly on their oxidation state. Monovalent and pentavalent ions were not extractable. In divalent metal extraction, Ca(II) (ionic radius: 100 pm) showed the highest D value and the D decreased with increase or decrease of the ionic radius. Tri- and tetravalent ions with ionic radii of 87-113 pm and 83-94 pm, respectively, showed the D over 1000, and the extracted chemical forms were determined to be M(TODGA)
-dodecane was investigated and discussed concerning distribution ratios (D) of metal ions and their ionic radii. The extractability for elements depends strongly on their oxidation state. Monovalent and pentavalent ions were not extractable. In divalent metal extraction, Ca(II) (ionic radius: 100 pm) showed the highest D value and the D decreased with increase or decrease of the ionic radius. Tri- and tetravalent ions with ionic radii of 87-113 pm and 83-94 pm, respectively, showed the D over 1000, and the extracted chemical forms were determined to be M(TODGA) (NO
(NO )
)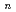 or M(TODGA)
or M(TODGA) (NO
(NO )
)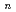 (
( =3 or 4). The oxonium ions with higher oxidation states than tetravalent one forms the complicated metal-complex with TODGA, and the number of TODGA associated in those extraction reactions was confirmed to be 2 at most.
=3 or 4). The oxonium ions with higher oxidation states than tetravalent one forms the complicated metal-complex with TODGA, and the number of TODGA associated in those extraction reactions was confirmed to be 2 at most.
Sasaki, Yuji; Choppin, G. R.*
Journal of Radioanalytical and Nuclear Chemistry, 246(2), p.267 - 273, 2000/11
Times Cited Count:38 Percentile:89.75(Chemistry, Analytical)no abstracts in English
Sasaki, Yuji
Bunseki Kagaku, 49(3), p.161 - 168, 2000/03
Times Cited Count:1 Percentile:4.58(Chemistry, Analytical)no abstracts in English
Sasaki, Yuji; Watanabe, Kazuo
JAERI-Research 99-005, 21 Pages, 1999/01
no abstracts in English
Sasaki, Yuji; G.R.Choppin*
Radiochimica Acta, 80(2), p.85 - 88, 1998/00
no abstracts in English
Sasaki, Yuji; Choppin, G. R.*
Journal of Radioanalytical and Nuclear Chemistry, 222(1-2), p.271 - 274, 1997/00
Times Cited Count:15 Percentile:90.13(Chemistry, Analytical)no abstracts in English